Mitochondrial Dysfunction and Bioenergetic Instability in Cancer Pathogenesis: Systematic Review of Mitochondria-Targeted Therapeutic Approaches
Keywords:
Mitochondrial Dysfunction, Bioenergetic Instability, Oxidative Phosphorylation, Reactive Oxygen Species, Cancer Therapy, Breast Cancer, Colorectal CancerAbstract
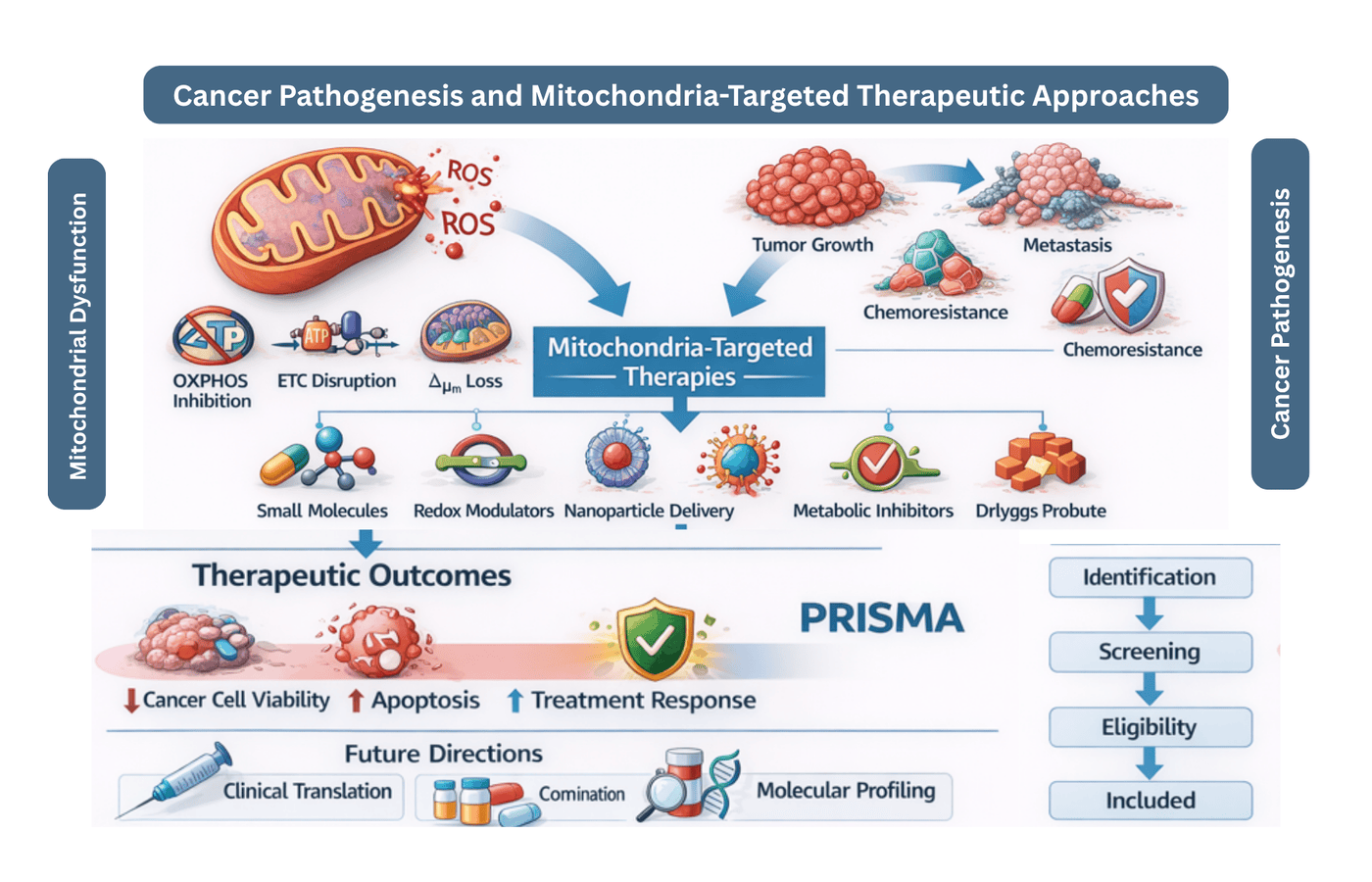
Background: Mitochondrial dysfunction and bioenergetic instability play key roles in cancer pathogenesis. This study aimed to systematically evaluate the role of mitochondria-targeted interventions in breast, lung, and colorectal cancers. Methods: This systematic review followed PRISMA guidelines 2020. Databases searched included PubMed/MEDLINE, Scopus, Web of Science, and Google Scholar, from 2020 to 2026. Experimental in vitro, in vivo, ex vivo, and clinical studies evaluating mitochondrial pathways or mitochondria-targeted interventions in breast, lung, and colorectal cancers were included, while reviews, editorials, case reports, conference abstracts, and non-English studies were excluded. Risk of bias was assessed using the SYRCLE tool, Cochrane Risk of Bias 2.0, and evidence certainty GRADE framework. Results: Twelve studies meet the inclusion criteria. Findings demonstrated that mitochondrial dysfunction, including OXPHOS inhibition, electron transport chain disruption, mitochondrial membrane depolarization, and ROS overproduction, contributed to tumor progression, chemoresistance, and metastasis. Mitochondria-targeted interventions reduced cancer cell viability, induced apoptosis, and improved treatment response. Risk of bias across studies was moderate, and the certainty of evidence was determined based on the GRADE approach. Conclusion: This review highlights the critical role of mitochondrial dysfunction in cancer progression and the potential of mitochondria-targeted therapies. Future research should focus on clinical translation, combination therapies, and molecular profiling to optimize patient-specific mitochondrial-based interventions.
Downloads
References
1. Napolitano G, Fasciolo G, Venditti P. Mitochondrial management of reactive oxygen species. Antioxidants (Basel). 2021;10(11):1824. https://doi.org/10.3390/antiox10111824
2. Liu Y, Sun Y, Guo Y, Shi X, Chen X, Feng W, et al. An overview: the diversified role of mitochondria in cancer metabolism. Int J Biol Sci. 2023;19(3):897–915. https://doi.org/10.7150/ijbs.81609
3. Xi Y, Xu P. Global colorectal cancer burden in 2020 and projections to 2040. Transl Oncol. 2021;14(10):101174.https://doi.org/10.1016/j.tranon.2021.101174
4. Yue SW, Liu HL, Su HF, et al. m6A-regulated tumor glycolysis: new advances in epigenetics and metabolism. Mol Cancer. 2023;22(1):137. https://doi.org/10.1186/s12943-023-01841-8
5. Van der Merwe M, van Niekerk G, Fourie C, du Plessis M, Engelbrecht AM. The impact of mitochondria on cancer treatment resistance. Cell Oncol (Dordr). 2021;44(1):983–995. https://doi.org/10.1007/s13402-021-00623-y
6. Chen F, Xue Y, Zhang W, Zhou H, Zhou Z, Chen T, et al. The role of mitochondria in tumor metastasis and advances in mitochondria-targeted cancer therapy. Cancer Metastasis Rev. 2024;43(1):1419–1443. https://doi.org/10.1007/s10555-024-10211-9
7. Avolio R, Matassa DS, Criscuolo D, Landriscina M, Esposito F. Modulation of mitochondrial metabolic reprogramming and oxidative stress to overcome chemoresistance in cancer. Biomolecules. 2020;10(1):135. https://doi.org/10.3390/biom10010135
8. Kozakiewicz P, Grzybowska-Szatkowska L, Ciesielka M, Rzymowska J. The role of mitochondria in carcinogenesis. Int J Mol Sci. 2021;22(10):5100. https://doi.org/10.3390/ijms22105100
9. Zhang L, Zhang W, Li Z, et al. Mitochondria dysfunction in CD8+ T cells as an important contributing factor for cancer development and a potential target for cancer treatment: a review. J Exp Clin Cancer Res. 2022;41(1):227. https://doi.org/10.1186/s13046-022-02439-6
10. MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372(8284): n71. https://doi.org/10.1136/bmj.n71
11. Hooijmans CR, Rovers MM, de Vries RB, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE's risk of bias tool for animal studies. BMC Med Res Methodol. 2014; 14(1):43. https://doi.org/10.1186/1471-2288-14-43
12. Nejadghaderi SA, Balibegloo M, Rezaei N. The Cochrane risk of bias assessment tool 2 (RoB 2) versus the original RoB: a perspective on the pros and cons. Health Sci Rep. 2024;7(6):e2165. https://doi.org/10.1002/hsr2.2165
13. Shen Y, Yang Y, Wang Z, Lin W, Feng N, Shi M, et al. Coptisine exerts anti-tumour effects in triple-negative breast cancer by targeting mitochondrial complex I. Br J Pharmacol. 2024;181(21):4262–4278. https://doi.org/10.1111/bph.16489
14. Ahn J, Kim OH, Jin S, Ryu JH, Lee D, Park WC, et al. Synergistic anticancer effects of mitochondria-targeting peptide combined with paclitaxel in breast cancer cells: a preclinical study. Ann Surg Treat Res. 2025;108(2):108–123. https://doi.org/10.4174/astr.2025.108.2.108
15. Modica-Napolitano JS, Clarke A, Nixdorf L, Shanahan B, Iacovella N, Reyes C, et al. Investigating the in vitro mitochondria-mediated anticancer activity of the plant metabolite ursolic acid. Int J Mol Sci. 2026;27(4):2067. https://doi.org/10.3390/ijms27042067
16. Kapan E, Uslu C, Arab H, Ahmed L, Ali R, Tereshchenkov AG, et al. Toward mitochondrial targeting of resistant triple-negative breast cancer using triphenylphosphonium-conjugated antimicrobial peptides. ACS Pharmacol Transl Sci. 2025;8(11):4159–4171. https://doi.org/10.1021/acsptsci.5c00563
17. Zhang C, Zhao X, Li F, Qin J, Yang L, Yin Q, et al. Integrating single-cell and multi-omic approaches reveals Euphorbiae Humifusae Herba-dependent mitochondrial dysfunction in non-small-cell lung cancer. J Cell Mol Med. 2024;28(10):e18317. https://doi.org/10.1111/jcmm.18317
18. Wang H, Shi W, Zeng D, Li X, Chen Y, Liu Q, et al. pH-activated, mitochondria-targeted, and redox-responsive delivery of paclitaxel nanomicelles to overcome drug resistance and suppress metastasis in lung cancer. J Nanobiotechnol. 2021;19(1):152. https://doi.org/10.1186/s12951-021-00895-4
19. Shameem M, Bagherpoor AJ, Nakhi A, Dosa P, Georg G, Kassie F. Mitochondria-targeted metformin (Mitomet) inhibits lung cancer in cellular models and in mice by enhancing the generation of reactive oxygen species. Mol Carcinog. 2023;62(11):1619–1629. https://doi.org/10.1002/mc.23603
20. Zhang Q, Xiong D, Pan J, Wang Y, Hardy M, Kalyanaraman B, et al. Chemoprevention of lung cancer with a combination of mitochondria-targeted compounds. Cancers. 2022;14(10):2538. https://doi.org/10.3390/cancers14102538
21. Wang I, Zhang L, Xin H, Guo Y, Zhu B, Su L, et al. Mitochondria-targeting folic acid-modified nanoplatform based on mesoporous carbon and a bioactive peptide for improved colorectal cancer treatment. Acta Biomater. 2022;152(1):453–472. https://doi.org/10.1016/j.actbio.2022.08.071
22. Wang X, Xu Z, Wang J, Wu C, Zhang L, Qian C, et al. A mitochondria-targeted biomimetic nanomedicine capable of reversing drug resistance in colorectal cancer through mitochondrial dysfunction. Adv Sci (Weinh). 2025;12(13):e2410630. https://doi.org/10.1002/advs.202410630
23. Fu Y, Ye Y, Zhu G, Xu Y, Sun J, Wu H, et al. Resveratrol induces human colorectal cancer cell apoptosis by activating the mitochondrial pathway via increasing reactive oxygen species. Mol Med Rep. 2021;23(3):170. https://doi.org/10.3892/mmr.2020.11809
24. Mathur S, Srivastava P, Srivastava A, et al. Regulation of metastatic potential by drug repurposing and mitochondrial targeting in colorectal cancer cells. BMC Cancer. 2024;24(1):323. https://doi.org/10.1186/s12885-024-12064-5
25. Zorov DB, Juhaszova M, Sollott SJ. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev. 2014;94(3):909-950. https://doi.org/10.1152/physrev.00026.2013
26.Vasan K, Werner M, Chandel NS. Mitochondrial metabolism as a target for cancer therapy. Cell Metab. 2020;32(3):341-352. https://doi.org/10.1016/j.cmet.2020.06.019
27. Raimondi V, Ciccarese F, Ciminale V. Oncogenic pathways and the electron transport chain: a dangerous liaison. Br J Cancer. 2020;122(2):168-181. https://doi.org/10.1038/s41416-019-0651-y
28. Osman AMA, Arabi AA. Targeting mitochondrial complexes for cancer therapy. Biochem Pharmacol. 2026;247(1):117695. https://doi.org/10.1016/j.bcp.2026.117695
29. Hill BG, Benavides GA, Lancaster JR Jr, Ballinger S, Dell'Italia L, Jianhua Z, et al. Integration of cellular bioenergetics with mitochondrial quality control and autophagy. Biol Chem. 2012;393(12):1485–1512. https://doi.org/10.1515/hsz-2012-0198
30. Farhana A, Alsrhani A, Khan YS, Rasheed Z. Cancer bioenergetics and tumor microenvironments—enhancing chemotherapeutics and targeting resistant niches through nanosystems. Cancers (Basel). 2023;15(15):3836. https://doi.org/10.3390/cancers15153836
31. Zhao Y, Ye X, Xiong Z, Ihsan A, Ares I, Martínez M, et al. Cancer metabolism: the role of ROS in DNA damage and induction of apoptosis in cancer cells. Metabolites. 2023;13(7):796. https://doi.org/10.3390/metabo13070796
32. Shimura T. Mitochondrial signaling pathways associated with DNA damage responses. Int J Mol Sci. 2023;24(7):6128. https://doi.org/10.3390/ijms24076128
33. Liu CJ, Wang LK, Tsai FM. The application and molecular mechanisms of mitochondria-targeted antioxidants in chemotherapy-induced cardiac injury. Curr Issues Mol Biol. 2025;47(3):176. https://doi.org/10.3390/cimb47030176
34. Ghosh P, Vidal C, Dey S, Zhang L. Mitochondria targeting as an effective strategy for cancer therapy. Int J Mol Sci. 2020;21(9):3363. https://doi.org/10.3390/ijms21093363
35. Ngoi NYL, Choong C, Lee J, Bellot G, Wong AL, Goh BC, et al. Targeting mitochondrial apoptosis to overcome treatment resistance in cancer. Cancers (Basel). 2020;12(3):574. https://doi.org/10.3390/cancers12030574
36. Lei ZN, Tian Q, Teng QX, Wurpel JND, Zeng L, Pan Y, et al. Understanding and targeting resistance mechanisms in cancer. MedComm. 2023;4(3):e265. https://doi.org/10.1002/mco2.265
37. Pyrczak-Felczykowska A, Herman-Antosiewicz A. Modification in structures of active compounds in anticancer mitochondria-targeted therapy. Int J Mol Sci. 2025;26(3):1376. https://doi.org/10.3390/ijms26031376
38. Merlin JPJ, Crous A, Abrahamse H. Combining photodynamic therapy and targeted drug delivery systems: enhancing mitochondrial toxicity for improved cancer outcomes. Int J Mol Sci. 2024;25(19):10796. https://doi.org/10.3390/ijms251910796
39. Tan EW, Singh SK, Dua K, Gupta G, Lee WL, Wong RSY, et al. Cancer stem cells: mitochondria signalling pathway and strategies for therapeutic interventions. Mol Biol Rep. 2025;52(1):671. https://doi.org/10.1007/s11033-025-10748-0
40. Chen G, Wang F, Trachootham D, Huang P. Preferential killing of cancer cells with mitochondrial dysfunction by natural compounds. Mitochondrion. 2010;10(6):614–625. https://doi.org/10.1016/j.mito.2010.08.001