Oxidative Stress, Mitochondrial Dysfunction, and Protein Misfolding Across Alzheimer’s, Parkinson’s, and Huntington’s Disease: A Systematic Review on Biomolecular Therapeutic Interventions
Keywords:
Oxidative Stress, Mitochondria, Protein Folding, Neuroprotection, Alzheimer Disease, Parkinson Disease, Huntington DiseaseAbstract
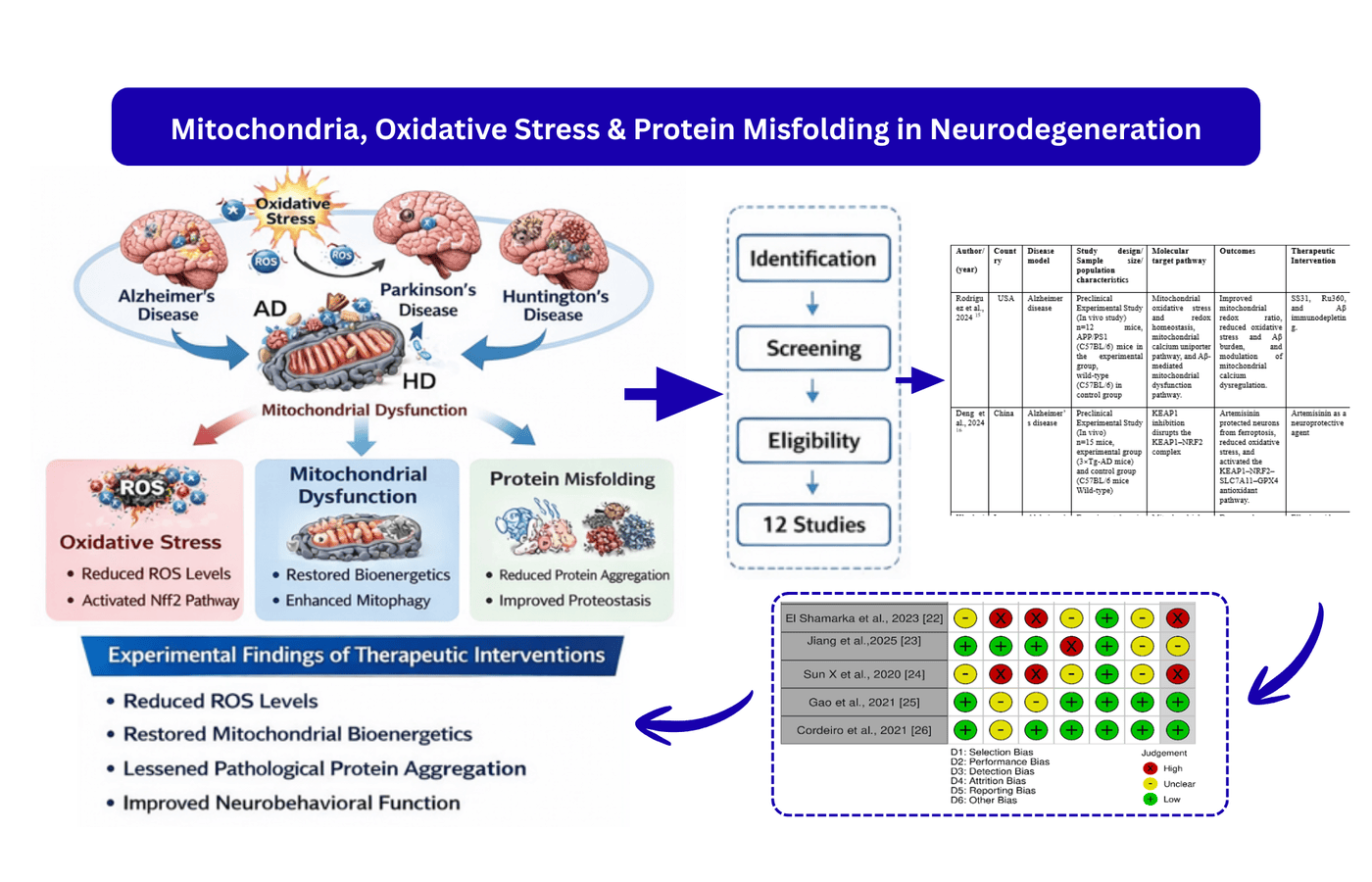
Background: Oxidative stress, mitochondrial dysfunction, and protein misfolding are some of the key and interrelated mechanisms in major neurodegenerative disorders. The purpose of this systematic review was to assess biomolecular therapeutic interventions of these pathways in the models of Alzheimer’s disease (AD), Parkinson’s disease (PD), and Huntington’s disease (HD). Methods: This is a systematic review that follow PRISMA 2020 guidelines. The searches took place up to from 2020 to 2026 in PubMed, Scopus, Web of Science, and Google Scholar. Experimental in vitro, in vivo, and ex vivo studies on biomolecular interventions that examine oxidative stress, mitochondrial dysfunction, or protein misfolding were accepted and reviews, editorials, conference abstracts, and non-English articles had been excluded. Screening and data extraction were done by two independent reviewers. The risk of bias was determined with the help of SYRCLE Risk of Bias tool of animal studies and Cochrane Risk of Bias 2.0 tool of in vitro studies and certainty of evidence was measured with the help of GRADE framework. Results: Twelve studies that passed the inclusion criteria were included following screening and eligibility. In disease models, interventions continually lowered the level of reactive oxygen species, restored mitochondrial bioenergetics, triggered Nrf2-mediated antioxidant response, lessened pathological protein aggregation, and neurobehavioral function. The risk of bias was low to moderate. Conclusion: The approach of the mitochondrial redox imbalance and proteostasis is a promising cross-disease treatment of neurodegeneration. To enhance the study further, future studies are needed to concentrate on standard experimental designs and clinical validation.
Downloads
References
1. Alqahtani T, Deore SL, Kide AA, Shende BA, Sharma R, Chakole RD, et al. Mitochondrial dysfunction and oxidative stress in Alzheimer's disease, Parkinson's disease, Huntington's disease and amyotrophic lateral sclerosis—an updated review. Mitochondrion.2023;71(6):83–92. https://doi.org/10.1016/j.mito.2023.05.007
2. Yang HM. Mitochondrial dysfunction in neurodegenerative diseases. Cells. 2025;14(4):276. https://doi.org/10.3390/cells14040276
3. Houldsworth A. Role of oxidative stress in neurodegenerative disorders: a review of reactive oxygen species and prevention by antioxidants. Brain Commun. 2024;6(1): fcad356. https://doi.org/10.1093/braincomms/fcad356
4. Zhang Y, Li XW, Zhang Y, Li X. Advances in research on mitochondrial dysfunction in neurodegenerative diseases. J Neurol. 2025;272(5):364. https://doi.org/10.1007/s00415-025-13101-4
5. Guan Y, Li L, Yang R, Lu Y, Tang J. Targeting mitochondria with natural polyphenols for treating Neurodegenerative Diseases: a comprehensive scoping review from oxidative stress perspective. J Transl Med. 2025;23(1):572. https://doi.org/10.1186/s12967-025-06605-0
6. Wong JH, Lai KO, Barron AM. Unlocking therapies for neurodegeneration: targeting mitochondrial dysfunction and oxidative stress in Alzheimer’s disease. Curr Opin Physiol. 2026; 47:100874. https://doi.org/10.1016/j.cophys.2025.100874
7. Toader C, Tataru CP, Munteanu O, Serban M, Covache-Busuioc RA, Ciurea AV, et al. Decoding neurodegeneration: a review of molecular mechanisms and therapeutic advances in Alzheimer’s, Parkinson’s, and ALS. Int J Mol Sci. 2024;25(23):12613. https://doi.org/10.3390/ijms252312613
8. Moselhy OA, Abdel-Aziz N, El-Bahkery A, Moselhy SS, Ibrahim EA. Curcumin nanoparticles alleviate brain mitochondrial dysfunction and cellular senescence in γ-irradiated rats. Sci Rep. 2025;15(1):3857. https://doi.org/10.1038/s41598-025-87635-y
9. Islam MT. Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol Res. 2017;39(1):73–82. https://doi.org/10.1080/01616412.2016.1251711
10. Meng K, Jia H, Hou X, Zhu Z, Lu Y, Feng Y, et al. Mitochondrial dysfunction in neurodegenerative diseases: mechanisms and corresponding therapeutic strategies. Biomedicines. 2025;13(2):327. https://doi.org/10.3390/biomedicines13020327
11. Wadan AH, Shaaban AH, El-Sadek MZ, Mostafa SA, Moshref AS, El-Hussein A, et al. Mitochondrial-based therapies for neurodegenerative diseases: a review of the current literature. Naunyn-schmiedeberg's Arch Pharmacol. 2025;398(1):11357–11386. https://doi.org/10.1007/s00210-025-04014-0
12. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372(8284): n71. https://doi.org/10.1136/bmj.n71
13. Hooijmans CR, Rovers MM, de Vries RB, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE's risk of bias tool for animal studies. BMC Med Res Methodol. 2014; 14(1):43. https://doi.org/10.1186/1471-2288-14-43
14. Nejadghaderi SA, Balibegloo M, Rezaei N. The Cochrane risk of bias assessment tool 2 (RoB 2) versus the original RoB: a perspective on the pros and cons. Health Sci Rep. 2024;7(6):e2165. https://doi.org/10.1002/hsr2.2165
15. Calvo-Rodriguez M, Kharitonova EK, Snyder AC, Hou SS, Sanchez-Mico MV, Das S, et al. Real-time imaging of mitochondrial redox reveals increased mitochondrial oxidative stress associated with amyloid β aggregates in vivo in a mouse model of Alzheimer’s disease. Mol Neurodegener. 2024;19(1):6. https://doi.org/10.1186/s13024-024-00702-2
16. Deng PX, Silva M, Yang N, Wang Q, Meng X, Ye KQ, et al. Artemisinin inhibits neuronal ferroptosis in Alzheimer's disease models by targeting KEAP1. Acta Pharmacol Sin. 2025;46(2):326–337. https://doi.org/10.1038/s41401-024-01378-6
17. Khodaei F, Namavar MR, Soutodeh N, Hadipour A, Asadi F, Khoshnoud MJ, et al. Mitochondrial protective effects of ellagic acid in a rat model of sporadic Alzheimer’s disease induced by STZ. Sci Rep. 2026;16(1):3498. https://doi.org/10.1038/s41598-025-31867-5
18. Chen H, Li N, Liu N, Zhu H, Ma C, Ye Y, et al. Photobiomodulation modulates mitochondrial energy metabolism and ameliorates neurological damage in an APP/PS1 mouse model of Alzheimer’s disease. Alz Res Ther. 2025;17(1):72. https://doi.org/10.1186/s13195-025-01714-w
19. Eo H, Yu SH, Choi Y, Kim Y, Kang YC, Lee H, et al. Mitochondrial transplantation exhibits neuroprotective effects and improves behavioral deficits in an animal model of Parkinson’s disease. Neurotherapeutics. 2024;21(4): e00355. https://doi.org/10.1016/j.neurot.2024e00355
20. Kim S, Choi JG, Kim SW, Park SC, Kang Y R, Park Ds, et al. Inhibition of α synuclein aggregation by MT101 5 is neuroprotective in mouse models of Parkinson’s disease. Biomed Pharmacother. 2022; 154:113637. https://doi.org/10.1016/j.biopha.2022.113637
21. Liu M, Zuo S, Guo X, Peng J, Xing Y, et al. The study of overexpression of Peroxiredoxin-2 reduces MPP+-induced toxicity in the cell model of Parkinson's disease. Neurochem Res. 2023;48(7):2129–2137. https://doi.org/10.1007/s11064-023-03880-5
22. El Shamarka ME, Abdel Salam OM, Shafee N, Zeidan HM. Curcumin modulation of L dopa and rasagiline induced neuroprotection in rotenone model of Parkinson’s disease. Iran J Basic Med Sci. 2023;26(2):139–147. https://doi.org/10.22038/IJBMS.2022.61687.13650
23. Jiang Z, Liu D, Li T, Gai C, Xin D, Zhao Y, et al. Hydrogen sulfide reduces oxidative stress in Huntington’s disease via Nrf2. Neural Regen Res. 2025;20(6):1776–1788. https://doi.org/10.4103/NRR.NRR D 23 01051
24. Sun X, Zhu J, Sun X, Ji M, Yu XL, Liu R. Ellagic acid rescues motor and cognitive deficits in R6/2 mouse model of Huntington’s disease by lowering mutant huntingtin protein. Food Funct. 2020;11(2):1334-1348. https://doi.org/10.1039/C9FO02131K
25. Pérez Arancibia R, Ordoñez JL, Rivas A, Pihán P, Sagredo A, Ahumada U, et al. A phenolic rich extract from Ugni molinae berries reduces abnormal protein aggregation in a cellular model of Huntington’s disease. PLoS One. 2021;16(7): e0254834. https://doi.org/10.1371/journal.pone.0254834
26. Cordeiro LM, Soares M, Silva A, Machado M, Obetine Baptista F, Silveira T, et al. Neuroprotective effects of rutin on ASH neurons in Caenorhabditis elegans model of Huntington’s disease. Nutr Neurosci. 2022; 25(11):1–14. https://doi.org/10.1080/1028415X.2021.1956254
27. Sweeney P, Park H, Baumann M, Dunlop J, Frydman J, Kopito R, et al. Protein misfolding in neurodegenerative diseases: implications and strategies. Transl Neurodegener. 2017; 6(1):6. https://doi.org/10.1186/s40035-017-0077-5
28. Wang W, Zhao F, Ma X, Perry G, Zhu X. Mitochondria dysfunction in Alzheimer’s disease. Neurosci Bull. 2020;36:220–232. https://doi.org/10.1007/s12264-019-00465-3
29. Angelova PR, Abramov AY. Role of mitochondrial ROS in the brain: from physiology to neurodegeneration. FEBS Lett. 2020;594:2370–2384. https://doi.org/10.1002/1873-3468.13714
30. Wen P, Sun Z, Gou F, Wang J, Fan Q, Zhao D, et al. Oxidative stress and mitochondrial impairment: key drivers in neurodegenerative disorders. Ageing Res Rev. 2025;104:102667. https://doi.org/10.1016/j.arr.2025.102667
31. Yusuf JA, Akanbi ST, Ijibadejo MS, Adebayo OS, Okoro CA, Bello OA, et al. A review of mitochondrial dysfunction, pathophysiology and therapeutic prospects in neurodegenerative diseases. Discov Neurosci. 2025;20(1):25. https://doi.org/10.1186/s13064-025-00223-8
32. Hou Y, Dan X, Babbar M, et al. Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol. 2023;19(1):608–624. https://doi.org/10.1038/s41582-023-00802-0
33. Percie du Sert N, Hurst V, Ahluwalia A, Alam S, Avey MT, Baker M, et al. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. Br J Pharmacol. 2020;177(16):3617–3624. https://doi.org/10.1111/bph.15193
34. Patel N, Satapathy T, Sahu P, Satapathy A, Bhardwaj SK, Satapathy A, et al. Molecular mechanisms of mitochondrial dysfunction in neurodegenerative diseases: pharmacological targets and therapeutic advances. J Drug Deliv Ther. 2025;15(10):272–294. https://doi.org/10.22270/jddt.v15i10.7424
35. Kallur S, Suryawanshi A, Utarade A, Kandalkar P, Morde R, Bhagwat A, et al. Oxidative stress and neurodegenerative diseases: exploring natural antioxidants for therapeutic potential. IP Int J Compr Adv Pharmacol. 2023;8(3):149–158. https://doi.org/10.18231/j.ijcaap.2023.026
36. Krsek A, Baticic L. Nanotechnology-driven therapeutic innovations in neurodegenerative disorders: a focus on Alzheimer’s and Parkinson’s disease. Future Pharmacol. 2024;4(2):352–379. https://doi.org/10.3390/futurepharmacol4020020
37. Chopade P, Chopade N, Zhao Z, Mitragotri S, Liao R, Chandran Suja V. Alzheimer’s and Parkinson’s disease therapies in the clinic. Bioeng Transl Med. 2022;8(1):e10367. https://doi.org/10.1002/btm2.10367
38. Gadhave DG, Sugandhi VV, Jha SK, Nangare SN, Gupta G, Singh SK, et al. Neurodegenerative disorders: mechanisms of degeneration and therapeutic approaches with their clinical relevance. Ageing Res Rev. 2024;99(1):102357. https://doi.org/10.1016/j.arr.2024.102357
39. Arimon M, Grimminger V, Sanz F, Lashuel HA. Hsp104 targets multiple intermediates on the amyloid pathway and suppresses the seeding capacity of fibrils and protofibrils of Aβ. J Mol Biol. 2008;384(5):1157–1173. https://doi.org/10.1016/j.jmb.2008.09.019
40. Oueslati A, Paleologou KE, Schneider BL, Aebischer P, Lashuel HA. Mimicking phosphorylation at serine 87 inhibits the aggregation of human alpha-synuclein and protects against its toxicity in a rat model of Parkinson’s disease. J Neurosci. 2012;32(5):1536–1544. https://doi.org/10.1523/JNEUROSCI.3781-11.2012