Plants Phytochemicals: Recovering Cancerous Cells
Keywords:
Phytochemicals, Neoplasms, Signal TransductionAbstract
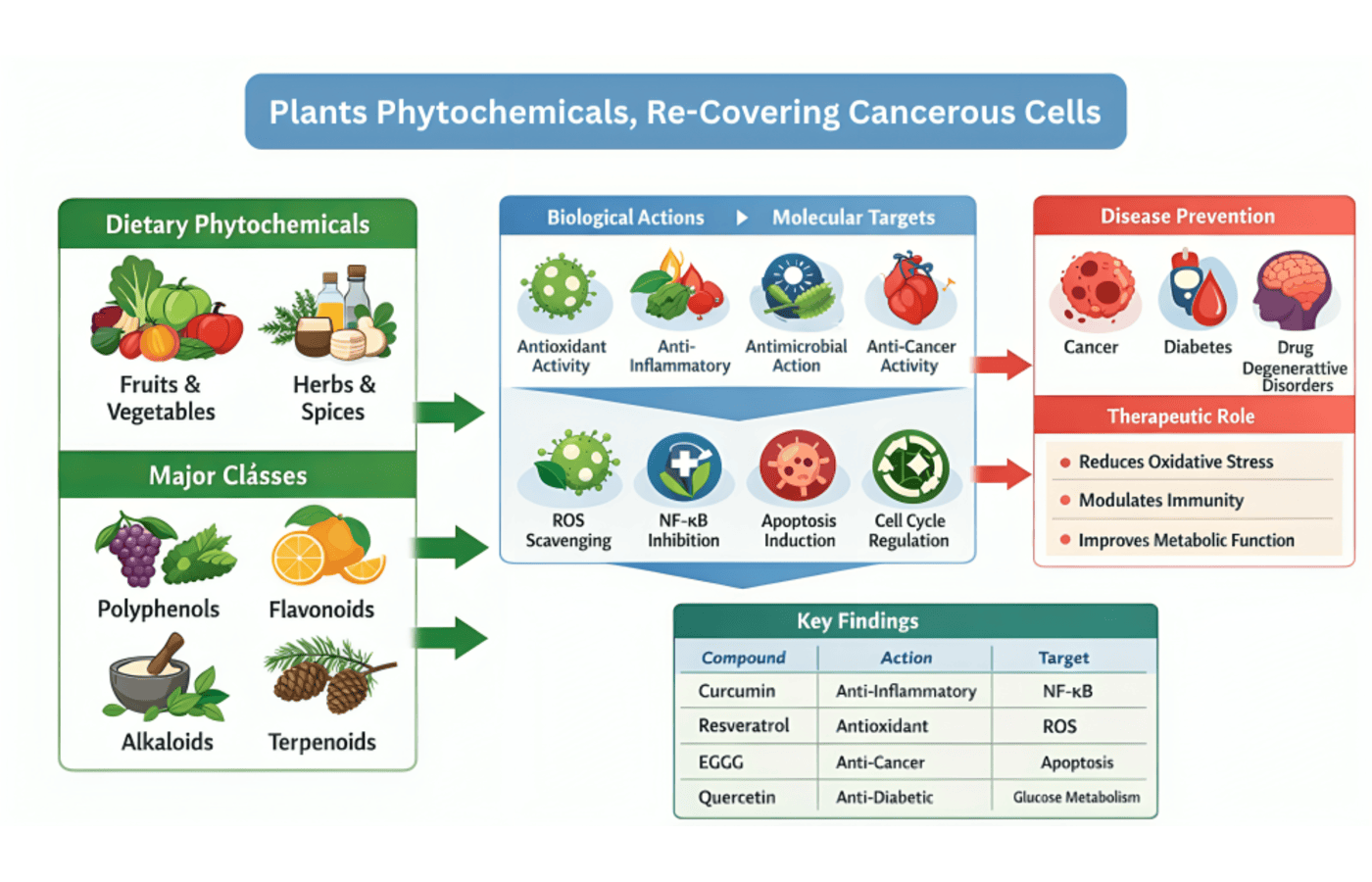
Cancer is a multifactorial disease of mammoth burden of global scale and the second major cause of death worldwide where millions of new cases are diagnosed each year 1. The cancer characteristics, such as long-term proliferative signaling, apoptotic resistance, invasion and metastasis, and immune destruction, are multifaceted therapeutic targets that cannot be adequately addressed by conventional therapeutic modalities like surgery, radiotherapy, and chemotherapy. The growing body of ethnopharmacological and molecular studies has recently shifted the focus to phytochemicals, which are secondary plant metabolites, as promising therapeutic agents in the recovery and reprogramming of cancerous cells 2. Phytochemicals are structurally heterogeneous bioactive substances that are produced by plants as elements of their adaptive defense systems. These molecules represent only a wide chemical distribution that comprise of phenolic acids, flavonoids, stilbenes, lignans, terpenoids, alkaloids, saponins and organosulfur compounds 3. Their anticancer action is not limited to cytotoxicity, but phytochemicals have multifaceted molecular negotiations with oncogenic signaling pathways, epigenetic modulators, and tumor microenvironment elements, which collectively tip the biological balance between a malignant and a quiescent or an apoptotic cellular state 4. The increasing in vitro, in vivo and preclinical evidence favors the therapeutic interest of phytochemicals in a wide variety of cancers, such as breast, colorectal, lung, hepatocellular, and prostate malignancies 5.
Flavonoids and cell cycle arrest
Flavonoids are a type of anticancer phytochemicals that are one of the best-researched classes of phytochemicals. Quercetin, kaempferol, luteolin and epigallocatechin-3-gallate (EGCG) have shown the ability to block cell cycle at the G1/S and G2/M checkpoints by regulating cyclins and cyclin-dependent kinases 6. These polyphenolic substances induce the expression of tumor suppressor proteins like p53 and p21, which provide molecular states that are hostile to the uncontrolled cell division. The role of flavonoids in the regulation of long non-coding RNA expression that regulates proliferative gene networks in colorectal and lung cancer cells has also been explained in recent studies 7. At the same time, flavonoids reduce the expression of antiapoptotic proteins such as Bcl-2 and Bcl-xL, and stimulate the induction of caspase-dependent apoptotic cascades. EGCG, a green tea extract, has shown specific ability to stimulate mitochondrial-pathway apoptosis in breast and prostate cancer by stimulating Bax and cytochrome c release 8. Notably, the most recent pharmacokinetic studies have discovered nanoencapsulation methods that can enhance the oral bioavailability of quercetin and kaempferol far beyond their established clinical translation shortcoming 9.
NF-B pathway and terpenoids inhibition
Another structurally diverse phytochemical group that has been shown to have anticancer activity is terpenoids. Triterpenoid including betulinic acid and ursolic acid induce apoptosis in cancer-causing cells but not normal tissue, which provides significant therapeutic benefit relative to traditional cytotoxic therapy 10. Sesquiterpene lactones, such as parthenolide and synthetic analogues, have been identified to inhibit nuclear factor-kappa B (NF- kB) pathway, which is a supreme controller of tumour survival, inflammatory crosstalk, and chemotherapy resistance. A study carried out in 2021 showed that parthenolide was able to sensitize triple-negative breast cancer cells to doxorubicin by disrupting antiapoptotic signalling orchestrated by NF-KB 11. NF-KB suppression by phytochemical therapies thus suppresses the expression of pro-survival cytokines, matrix metalloproteinases, and angiogenic factors which promote tumour growth (Figure 1). Recent in vivo models have demonstrated impaired Ras-MAP kinase signaling and lowered tumor burden in pancreatic and colorectal cancer xenografts with limonene and perillyl alcohol, monocyclic monoterpenes derived by citrus peel, supporting the mechanistic diversity of terpenoid anticancer action 12.
PI3K/Akt/mTOR signaling axis modulation.
The PI3K/Akt/mTOR pathway is a common dysregulated signalling pathway in human tumors and enhances cell survival, proliferation and reorganization of cancerous cellular metabolism. A number of phytochemicals have been reported to be strong agonists of this pathway. Resveratrol is a stilbene present in grapes and red berries that suppresses phosphorylation of Akt and subsequent mTORC1 activation thus limiting protein production and causing autophagy-mediated death in cancer lines 13. The main bioactive curcuminoid of Curcuma longa, curcumin, also inhibits Akt and mTOR signaling and at the same time stimulates AMP-activated protein kinase, shifting the cancer cell metabolism towards catabolic signaling pathways that cannot support malignant growth 14. The formulation innovations such as phospholipid complexes and self-emulsifying drug delivery systems have facilitated the clinical translation of curcumin, and Phase II clinical trials have shown favorable safety profiles, as well as initial evidence of tumor marker reduction in colorectal cancer patients 15.
Phytochemicals and anti-angiogenic mechanisms
The angiogenic pathway of tumours which attracts new vascularity to meet its oxygen and nutrient needs is a key target of therapy in oncology. Genistein, epigallocatechin gallate and thymoquinone, phytochemicals have been observed to prevent vascular endothelial growth factor (VEGF) expression and receptor tyrosine kinase activity effectively starving tumours of the blood supply needed to grow and metastatically spread 16. Nigella sativa yields thymoquinone, which has been studied with great intensity, with several papers demonstrating its ability to suppress hypoxia-inducible factor-1alpha (HIF-1 alpha), the major transcriptional regulator of VEGF in hypoxic tumour cores 17. These antiangiogenic effects of plant bioactives are complementary to the already available anti-VEGF pharmacological agents and have been shown to add to the effect of known antiangiogenic agents in preclinical tumor models, implying future clinical protocols would be complementary with current antiangiogenic therapies 18.
Re-educating the tumor microenvironment
The tumour microenvironment (TME) is an immunosuppressive niche that comprises regulatory T cells, myeloid-derived suppressor cells, cancer-associated fibroblasts and polarized M2 macrophages and together they inhibit antitumour immune responses. Recently, phytochemicals have been identified to be regulators of the TME, with the ability to remodel immunosuppressive immune cells into cytotoxic phenotypes. Sulforaphane, an isothiocyanate of cruciferous vegetables activates M1 macrophage polarisation and increases dendritic cell maturation, and thus, rescues immunosurveillance in the tumour milieu 19. Recent studies have shown that the berberine and luteolin inhibit the activation of cancer-associated fibroblasts and lower the release of tumour-promoting growth factors such as transforming growth factor- 20 and stromal cell-derived factor-1 20.
Phytochemicals as epigenetic modulators
A central element of the process of carcinogenesis is epigenetic dysregulation, which entails altered DNA-methylated states, histone-altered states, and non-coding RNA. Phytochemicals also have significant epigenetic modulatory properties, which provides a mechanism of gene expression normalisation to enhance direct cytotoxicity. EGCG is a DNA methyltransferase (DNMT) inhibitor, which reactivates tumour suppressor gene silenced in colorectal and breast cancers by promoter hypermethylation. One study has shown that inhibition of DNMT3a by EGCG could restore DKK3 Wnt antagonist expression in hepatocellular carcinoma and inhibit the expansion of tumors in vivo significantly 21. Curcumin suppresses histone deacetylases (HDACs) and increases acetylation of tumour suppressor gene loci that stimulate their transcriptional reinstatement. Moreover, resveratrol stimulates the action of Sirtuin-1 (SIRT1), a class III HDAC, which alters the acetylation levels of p53 and NF-kB and shifts their action towards tumour suppressive and not protective action 22. Together, these epigenetic activities have indicated that phytochemicals might be especially useful at reversing oncogenic epigenomic programming which presently represents a growing clinical goal due to the emerging epigenetic cancer therapy landscape 23.
Hormonal control cancer and glucosinolates
The Glucosinolates and hydrolysis products of glucosinolates, especially the indole-3-carbinol (I3C) and 3,3'-diindolylmethane (DIM) produced by Brassica species, have proven pleiotropic anticancer properties with particular application against hormonally-driven cancer. I3C alters oestrogen receptor signalling, favours the 2-hydroxylation of estradiol to the more genotoxic 16alpha-hydroxylation pathway and suppresses the expression of the oncogene HER2/neu in breast cancer models 24. In a 2021 clinical pilot study, DIM supplementation in women with early-stage cervical intraepithelial neoplasia had been found to achieve histological regression in a considerable proportion of participants over placebo, giving the preclinical supportive evidence on the hypothesized mechanism translational credibility 25. These phytochemicals comprise a mechanistically dissimilar group of anticancer agents that have been specifically employed in breast, cervical, and endometrial carcinomas, in which the hormonal disturbance contributes to tumor growth. In the future, further randomized controlled trials should determine the clinical dosing and efficacy parameters of them 26.
CONCLUSION
Plant phytochemicals are scientifically sound and diverse in mechanism bioactive molecules that have considerable potential in the re-covering of cancerous cells. Phytochemicals interact with malignant cellular biology at multiple points of vulnerability concurrently, by modulating apoptotic cascades, oncogenic signaling pathways, angiogenesis, epigenetic programming, immune microenvironmental dynamics as well as drug resistance mechanisms. The available preclinical evidence has shown persuading anticancer activities in a wide range of cancers and phytochemical families. The findings are increasingly being confirmed by translational clinical trials, but issues relating to bioavailability, standardization, and pharmacokinetic optimization still need to be overcome. Implementation of phytochemical-based interventions into a multidisciplinary oncology care is likely to revolutionize the therapeutic outcomes, treatment-related toxicity, and survival rates in cancer patients.
Downloads
References
1. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229-263. https://doi.org/10.3322/caac.21834
2. Atanasov AG, Zotchev SB, Dirsch VM, Supuran CT. Natural products in drug discovery: advances and opportunities. Nat Rev Drug Discov. 2021;20(3):200-216. https://doi.org/10.1038/s41573-020-00114-z
3. Kumar S, Pandey AK. Chemistry and biological activities of flavonoids: an overview. ScientificWorldJournal. 2023;2023(1):162750. https://doi.org/10.1155/2023/162750
4. Gupta SC, Sung B, Kim JH, Prasad S, Li S, Aggarwal BB. Multitargeting by turmeric, the golden spice: from kitchen to clinic. Mol Nutr Food Res. 2023;57(9):1510-1528. https://doi.org/10.1002/mnfr.201200741
5. Newman DJ, Cragg GM. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J Nat Prod. 2020;83(3):770-803. https://doi.org/10.1021/acs.jnatprod.9b01285
6. Kopustinskiene DM, Jakstas V, Savickas A, Bernatoniene J. Flavonoids as anticancer agents. Nutrients. 2020;12(2):457. https://doi.org/10.3390/nu12020457
7. Bian Y, Wei J, Zhao C, Li G. Natural polyphenols targeting senescence: a novel prevention and therapy strategy for cancer. Int J Mol Sci. 2020;21(2):684. https://doi.org/10.3390/ijms21020684
8. Yang CS, Wang H, Sheridan ZP. Studies on prevention of obesity, metabolic syndrome, diabetes, cardiovascular disease and cancer by tea. J Food Drug Anal. 2018;26(1):1-13. https://doi.org/10.1016/j.jfda.2017.10.010
9. Nile SH, Baskar V, Selvaraj D, Nile A, Xiao J, Kai G. Nanotechnologies in food science: applications, recent trends, and future perspectives. Nano-Micro Lett. 2020;12(1):45. https://doi.org/10.1007/s40820-020-0383-9
10. Yin R, Li T, Tian JX, Xi P, Liu RH. Ursolic acid, a potential anticancer compound for breast cancer therapy. Crit Rev Food Sci Nutr. 2018;58(4):568-574. https://doi.org/10.1080/10408398.2016.1203755
11. Nakshatri H, Appaiah HN, Anjanappa M, D Gilley, H Tanaka, S Badve, et al. NF-κB-dependent and -independent epigenetic modulation using the novel anti-cancer agent DMAPT. Cell Death Dis. 2021(2);6:e1608. https://doi.org/10.1038/cddis.2021.1608
12. Saldanha SN, Tollefsbol TO. Synergistic methodologies in the study of phytochemical-mediated epigenetic cancer therapy. Methods Mol Biol. 2022(1);2423:241-256. https://doi.org/10.1007/978-1-0716-1952-0_15
13. Ko JH, Sethi G, Um JY, Shanmugam MK, Arfuso F, Kumar AP, et al. The role of resveratrol in cancer therapy. International journal of molecular sciences. Int J Mol Sci. 2017;18(12):2589. https://doi.org/10.3390/ijms18122589
14. Kunnumakkara AB, Harsha C, Banik K, Vikkurthi R, Sailo BL, Bordoloi D, et al. Is curcumin bioavailability a problem in humans: Lessons from clinical trials. Expert Opin Drug Metab Toxicol. 2019;15(9):705-733. https://doi.org/10.1080/17425255.2019.1650914
15. Salehi B, Stojanovic-Radic Z, Matejic J, Sharifi-Rad M, V. Anil N, Martins N, et al. The therapeutic potential of curcumin: a review of clinical trials. Eur J Med Chem. 2019;163(1):527-545. https://doi.org/10.1016/j.ejmech.2018.12.016
16. Siddiqui IA, Sanna V, Ahmad N, Sechi M, Mukhtar H. Resveratrol nanoformulation for cancer prevention and therapy. Ann N Y Acad Sci. 2016;1348(1):20-31. https://doi.org/10.1111/nyas.13118
17. Imran M, Rauf A, Khan IA, Shahbaz M, Qaisrani T, Fatmawati S, et al. Thymoquinone: a novel strategy to combat cancer: a review. Biomed Pharmacother. 2018(1);106:390-402. https://doi.org/10.1016/j.biopha.2018.06.159
18. Biswas P, Dey D, Biswas PK, Rahaman TI, Saha S, Parvez A, et al. A comprehensive analysis and anti-cancer activities of quercetin in ROS-mediated cancer and cancer stem cells. Int J Mol Sci. 2022;23(19):11746. https://doi.org/10.3390/ijms231911746
19. Yi T, Liu Z, Jia H, Liu Q, Peng J. Sulforaphane regulates macrophage M1/M2 polarization to attenuate macrophage-induced Caco-2 cell injury in an inflammatory environment. Iran J Immunol. 2024;21(1):37–52. https://doi.org/10.22034/iji.2024.98644.2580
20. Samad MA, Saiman MZ, Abdul Majid N, Karsani SA, Yaacob JS. Berberine inhibits telomerase activity and induces cell cycle arrest and telomere erosion in colorectal cancer cell line HCT 116. Molecules. 2021;26(2):376. https://doi.org/10.3390/molecules26020376
21. Lyu H, Huang J, He Z, Liu B. Epigenetic mechanism of survivin dysregulation in human cancer. Sci China Life Sci. 2018;61(7):808–814. https://doi.org/10.1007/s11427-017-9230-2
22. Huang D, Cui L, Ahmed S, et al. An overview of epigenetic agents and natural nutrition products targeting DNA methyltransferase, histone deacetylases and microRNAs. Food Chem Toxicol. 2019;123(1):574-594. https://doi.org/10.1016/j.fct.2018.10.052
23. Cully M. Cancer: positive results for epigenetic drugs. Nat Rev Drug Discov. 2022;21(6):400-401. https://doi.org/10.1038/d41573-022-00076-4
24. Thomson CA, Ho E, Strom MB. Chemopreventive properties of 3,3’-diindolylmethane in breast cancer: evidence from experimental and human studies. Nutr Rev. 2016;74(7):432-443. https://doi.org/10.1093/nutrit/nuw010
25. Greenwald P, Anderson D, Nelson SA, Taylor PR. Clinical trials of vitamin and mineral supplements for cancer prevention. Am J Clin Nutr. 2020;75(7):1435S-1449S. https://doi.org/10.1093/ajcn/75.7.1435S
26. Bayat Mokhtari R, Homayouni TS, Baluch N, Morgatskaya E, Kumar S, Das B, et al. Combination therapy in combating cancer. Oncotarget. 2017;8(23):38022–38043. https://doi.org/10.18632/oncotarget.16723