Periodontal-Cardiometabolic Axis: How Oral Biology Shapes Critical Care Outcomes
Keywords:
Periodontal-Cardiometabolic Axis, Oral Biology, Critical Care, Advances in Medical SciencesAbstract
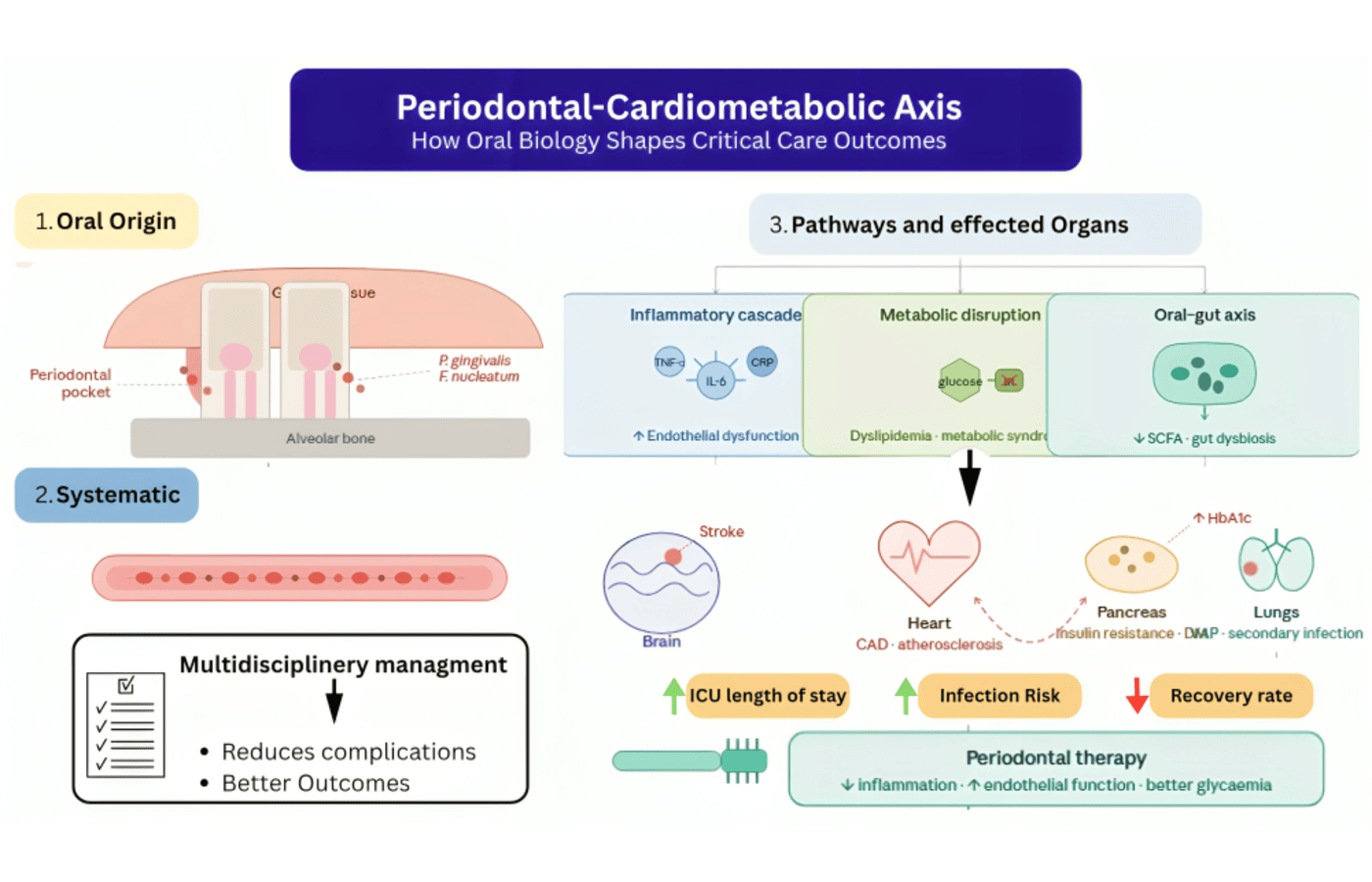
Periodontal disease is among the most prevalent chronic inflammatory diseases globally, that is characterized by unremitting dysbiosis of microbes and destruction of tooth supportive tissue 1. Periodontitis is traditionally thought of as a localized oral disease but today is being perceived as a systemic inflammatory disease with extensive health consequences. The damaged periodontal pocket epithelium offers a direct pathway of entry of oral bacteria and their toxic by-products into the bloodstream leading to recurrent occurrences of bacteremia and systemic immune response 2. This unending microbial challenge is central in influencing the so called periodontal-cardiometabolic axis. Chronic periodontal inflammation favors the release of systemic inflammatory mediators such as interleukin-6, tumor necrosis factor-a and C-reactive protein that are highly implicated in endothelial dysfunction and atherogenesis 3. These circulating mediators destroy vascular homeostasis, diminish nitric oxide levels of bioavailability and increase oxidative stress to form a biological environment where cardiovascular disease is favorable. Most recent molecular and epidemiological findings indicated that moderate to severe periodontitis patients have a greater number of systemic inflammatory indicators and are at a higher risk of coronary artery disease and ischemic stroke 4. Oral microbial by-products have direct effects on cardiometabolic pathways in addition to having effects on inflammatory signaling. The major periodontal pathogens like Porphyromonas gingivalis and Fusobacterium nucleatum have the capability of spreading to the bloodstream and engaging with the endothelial cells in the vascular beds, macrophages and platelets 5. Gram-negative oral bacteria lipopolysaccharides initiate Toll-like receptor signaling and nuclear factor-kB signaling and stimulate chronic low-grade inflammation and rapid atherosclerotic plaque 6. The mechanistic proof that these biological mechanisms have in support of the part of oral microbiology in cardiovascular pathology is substantiated.
The oral microbiome also interacts with the systemic metabolism in the oral-gut microbiota axis. New findings have shown that the gastrointestinal tract can be colonized by oral bacteria and restructure intestinal microbial colonies, which alters the generation of short-chain fatty acids structuring the bile acid metabolism and inflammatory metabolites 7. Such microbial crosstalk has been attributed to insulin resistance, dyslipidemia and metabolic syndrome, and thus, supports the correlation between periodontal disease and cardiometabolic dysfunction 8. A typical and two-way relationship is the case of diabetes mellitus and periodontal disease. Hyperglycemia affects the neutrophil activity, collagen metabolism and vascular integrity, making them the predisposing factors of periodontal disease and tissue erosion 9. Periodontal inflammation on the other hand enhances insulin resistance by maintaining systemic inflammatory load and disrupting the insulin signalling pathways 10. According to recent clinical data, a person with inadequately managed type 2 diabetes and active periodontitis is at higher risk of cardiovascular events and complications significantly than a metabolically healthy person 11.
These are the biological interactions that are especially pertinent to the cases of critical illness. Inflammation on a systemic scale, loss of control over the immune system and metabolic imbalance are significant prognostic variables in intensive care units. Periodontal disease has the potential to add to the cumulative inflammatory burden and could enhance inflammatory responses in sepsis, acute cardiovascular events and metabolic decompensation 12. The similar levels of C-reactive protein and pro-inflammatory cytokines found in severe periodontitis and severely ill patients indicate similar inflammatory pathways that may exacerbate organ dysfunction and slow down the recovery process 13. The translocation of oral microbial organisms can also impact the intensive care outcomes through making the patients vulnerable to secondary infections and maladaptive immune activities. The poor oral hygiene and decreased salivary flow in critically unstable patients encourages the growth of oral microbes, which can act as a source of respiratory and systemic pathogens 14. Despite the limited number of direct interventional trials, recent narrative and clinical reviews indicate that poor oral health correlates with longer hospitalization, increased risk of infection and adverse recovery trajectories on vulnerable hospitalized populations 15. Understanding of the periodontal-cardiometabolic axis has led to the focus of the significance of combined medical and dental treatment. The beneficial effects of periodontal therapy on endothelial function, as well as the decrease of systemic inflammatory markers, have demonstrated that oral inflammation management could potentially help to reduce cardiometabolic risks on a broader level 16.
Integrating oral health assessment into cardiometabolic and critical care interventions could thus be a significant and changeable practice to enhance patient outcome. In conclusion, cardiometabolic diseases are serious biological risks of periodontal disease through the process of chronic inflammation, microbial dysbiosis, and immune-metabolic interactions The existing literature shows that oral biology does have a significant role in the formation of cardiovascular risk, the control of diabetes, and possibly the results in intensive care. The introduction of periodontal care into multidisciplinary cardiometabolic and critical care systems has the potential to decrease systemic complications and aid in the long-term recovery of high-risk groups of patients.
Downloads
References
1. Cosmin Bida F, Curca FR, Lupusoru RV, et al. The systemic link between oral health and cardiovascular disease: contemporary evidence and mechanisms. Diseases. 2025 Nov;13(11):354. https://doi.org/10.3390/diseases13110354
2. Villoria GEM, Fischer RG, Tinoco EMB, Meyle J, Loos BG. Periodontal disease as a systemic condition. Periodontol 2000. 2024 Oct;96(1):7–19. https://doi.org/10.1111/prd.12499
3. Wang Z, Kaplan RC, Burk RD, Qi Q. The oral microbiota, microbial metabolites and immuno-inflammatory mechanisms in cardiovascular disease. Int J Mol Sci. 2024 Nov;25(22):12337. https://doi.org/10.3390/ijms252212337
4. Church L, Franks K, Medara N, Curkovic K, Singh B, Mehta J, et al. Impact of oral hygiene practices in reducing cardiometabolic risk, incidence, and mortality: a systematic review. International journal of environmental research and public health. Int J Environ Res Public Health. 2024 Oct;21(10):1319. https://doi.org/10.3390/ijerph21101319
5. Xu Q, Wang W, Li Y, et al. The oral–gut microbiota axis as a link in cardiometabolic diseases. NPJ Biofilms Microbiomes. 2025 Jan;11:11. https://doi.org/10.1038/s41522-025-00411-1
6. Zaman MS, Alam SMG, Razzaque MS. Oral hygiene and cardiovascular health. Hygiene. 2025 Apr;5(2):14. https://doi.org/10.3390/hygiene5020014
7. Li Y, Liu Y, Cui J, Zhu M, Wang W, Chen K, et al. Oral-gut microbial transmission promotes diabetic coronary heart disease. Cardiovascular Diabetology. Cardiovasc Diabetol. 2024 Apr;23(1):123. https://doi.org/10.1186/s12933-024-02217-y
8. Țica O, Romanul I, Ciavoi G, Pantea VA, Scrobota I, Șipoș L, et al. A clinical review of the connections between diabetes mellitus, periodontal disease, and cardiovascular pathologies. Biomedicines. 2025 Sep;13(9):2309. https://doi.org/10.3390/biomedicines13092309
9. Sanz M., Marco Del Castillo A., Jepsen S., et al. Periodontitis and diabetes: a two-way relationship. J Clin Periodontol. 2023 Jul;50(7):874-885. https://doi.org/10.1111/jcpe.13789
10. Song TJ, Jeon J, Kim J. Cardiovascular risks of periodontitis and oral hygiene indicators in patients with diabetes mellitus. Diabetes Metab J. 2021 Nov;47(6):101252. https://doi.org/10.1016/j.diabet.2021.101252
11. Temur I, Dönertas SN, Dönertaş AD, Arslan S, Temur KT, Magat G. Association of periodontal diseases with inflammatory markers and gestational diabetes: a case-control study. BMC Oral Health. 2025 Oct;25(1):1563. https://doi.org/10.1186/s12903-025-06929-9
12. An overlooked connection: oral health status in patients with chronic diseases. BMC Oral Health. 2025;25:5673. https://doi.org/10.1186/s12903-025-05673-4
13. Inoue S, Nakanishi N, Amaya F, Fujinami Y, Hatakeyama J, Hifumi T, et al. Post-intensive care syndrome: recent advances and future directions. Acute Med Surg. 2024 Jan;11(1):e929. https://doi.org/10.1002/ams2.929
14. Nakanishi N, Liu K, Hatakeyama J, Kawauchi A, Yoshida M, Sumita H, et al. Post-intensive care syndrome follow-up system after hospital discharge: a narrative review. J Intensive Care. 2024 Jan;12(1):2. https://doi.org/10.1186/s40560-023-00716-w
15. Ekong M, Monga TS, Daher JC, Sashank M, Soltani SR, Nwangene NL, et al. From the intensive care unit to recovery: managing post-intensive care syndrome in critically ill patients. Cureus. 2024 May;16(5):e61443. https://doi.org/10.7759/cureus.61443
16. Major ME, Dettling-Ihnenfeldt D, Ramaekers SP, Engelbert RH, van der Schaaf M. Feasibility of a home-based interdisciplinary rehabilitation program for patients with post-intensive care syndrome: the REACH study. Crit Care. 2021 Dec; 25:309. https://doi.org/10.1186/s13054-021-03709-z
Downloads
Published
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.

