Molecular Insights into Adipokines in Metabolic Syndrome: Implications for Novel Therapeutic Strategies
Keywords:
Metabolic Syndrome, Adipokines, Leptin, Adiponectin, Insulin Resistance, PolymorphismAbstract
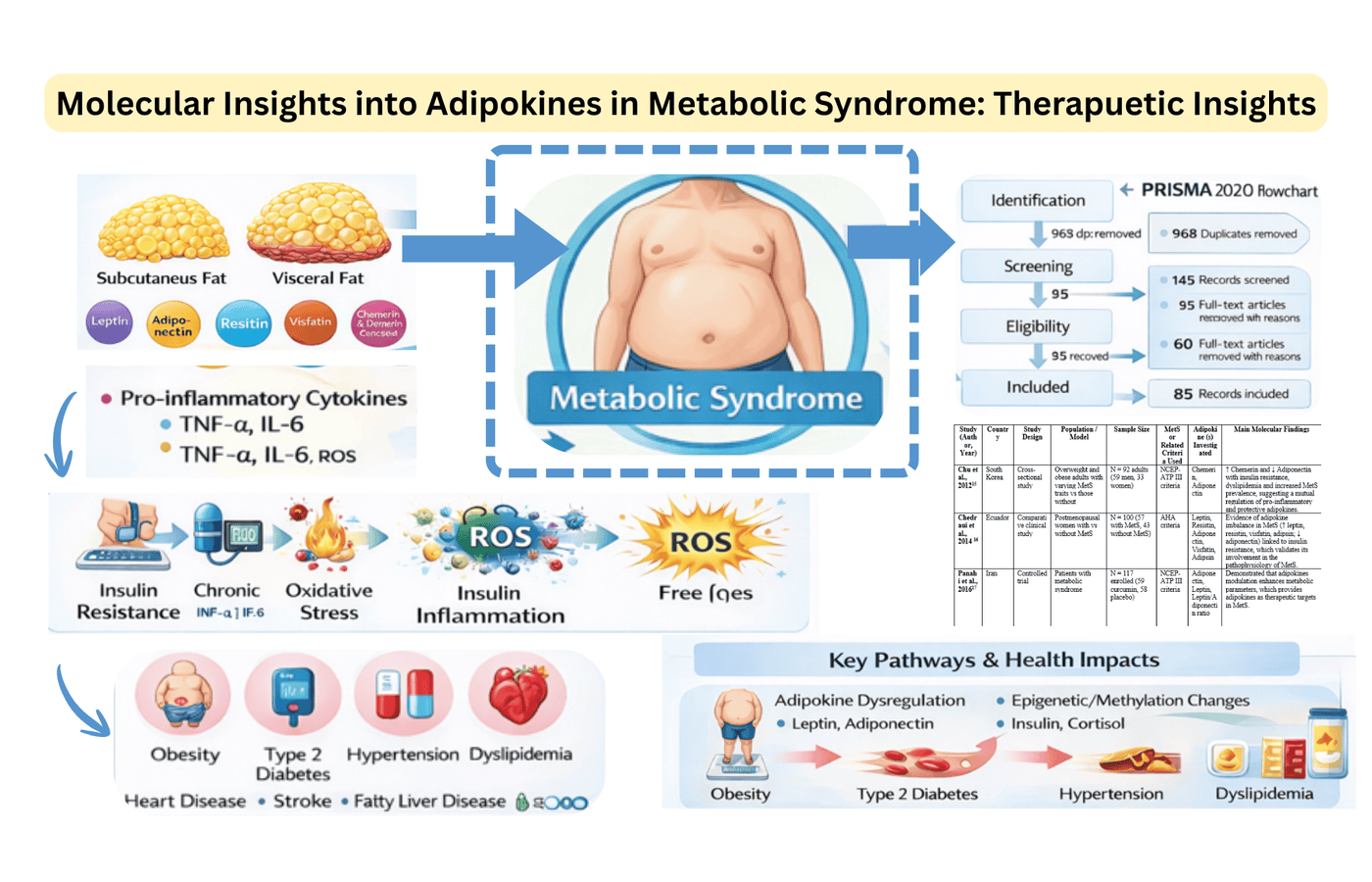
Background: Adipokines are bio-active peptides released by adipose tissue which control glucose and lipid metabolism, play important roles in pathophysiology of metabolic syndrome (MetS). The main objective of this systematic review was to collect molecular evidence of adipokine dysregulation in MetS and fully evaluate its clinical, genetic, and epigenetic implications. Methodology: Articles published since January 2014 to January 2026 were searched from different databases like PubMed, Scopus, Web of Science, and Google Scholar, by following PRISMA 2020 guidelines. Inclusion criteria were adults, MetS, and quantitative data on adipokines and their association with clinical or genetic variables. The quality was measured using the Newcastle-Ottawa Scale, ROBINS-I tool, and GRADE frameworks. Results: 12 out of 85 records met the inclusion criteria. MetS populations were always associated with an increase in pro-inflammatory adipokines (leptin, resistin, chemerin, visfatin) and a decrease in adiponectin, which are strictly linked to insulin resistance and inflammation. The severity of MetS was more strongly associated with visceral adipose tissue expression as compared to circulating levels alone. Adiponectin and leptin genetic variations (e.g., ADIPOQ, 73 CpG) and epigenetic changes (211 CpGs) controlled the expression of adipokines. The quality was quite average, with observational designs making it hard to make causal inferences. Conclusion: The dysregulation of adipokines is the core of MetS pathophysiology with tissue-specific expression and epigenetic regulation being the factors involved in metabolic dysfunction. Long-term, multi-omics longitudinal studies are necessary in the future to confirm therapeutic targets and allow individual interventions.
Downloads
References
1. Kim JE, Kim JS, Jo MJ, Cho E, Ahn SY, Kwon YJ, et al. The roles and associated mechanisms of adipokines in development of metabolic syndrome. Molecules. 2022;27(2):334. https://doi.org/10.3390/molecules27020334
2. Gupta A, Gupta P, Singh AK, Gupta V. Association of adipokines with insulin resistance and metabolic syndrome including obesity and diabetes. GHM Open. 2023;3(1):7–19. https://doi.org/10.35772/ghmo.2023.01004
3. Lehr S, Hartwig S, Sell H. Adipokines: A treasure trove for the discovery of biomarkers for metabolic disorders. Proteomics Clin Appl. 2012;6(1–2):91–101. https://doi.org/10.1002/prca.201100086
4. Helfer G, Wu Q-F. Chemerin: a multifaceted adipokine involved in metabolic disorders. J Endocrinol. 2018; 238(2):R79-R94. https://doi.org/10.1530/JOE-18-0174
5. Korac A, Srdic Galic B, Kalezic A, Stancic A, Otasevic V, Korac B, Jankovic A. Adipokine signatures of subcutaneous and visceral abdominal fat in normal weight and obese women with different metabolic profiles. Arch Med Sci. 2021;17(2):323–336. https://doi.org/10.5114/aoms.92118
6. Palit SP, Banerjee S, Dutta P, Ghosh S, Ray D, Mukherjee S, et al. A genetic analysis identifies a haplotype at the adiponectin locus associated with metabolic traits. Sci Rep. 2020;10(1):13045. https://doi.org/10.1038/s41598-020-59845-z
7. Pant R, Sharma S, Verma N, Singh P, Gupta R, Kumar A, et al. Epigenetic regulation of adipogenesis in adipose tissue dysfunction and metabolic disease. Front Cell Dev Biol. 2021;9:619888. https://doi.org/10.3389/fcell.2020.619888
8. Ibrahim MM. Subcutaneous and visceral adipose tissue: structural and functional differences. Obes Rev. 2010;11(1):11–18. https://doi.org/10.1111/j.1467-789X.2009.00623.x
9. Svensson H, Odén B, Edén S, Lönn M. Adiponectin, chemerin, cytokines, and dipeptidyl peptidase 4 are released from human adipose tissue in a depot dependent manner: an in vitro system including human serum albumin. BMC Endocr Disord. 2014;14(1):7. https://doi.org/10.1186/1472-6823-14-7
10. Fang MY, Liu SW, Kolatkar PR, Qi P, Liu YC, Sun F, et al. DNA methylation reshapes the expression of adipokines and adipose factors in white adipose tissues. Epigenetics Chromatin. 2025;18(1):71. https://doi.org/10.1186/s13072-025-00640-3
11. Kumari B, Yadav UCS. Adipokine Visfatin’s Role in Pathogenesis of Diabesity and Related Metabolic Derangements. Curr Mol Med. 2018;18(2):116-125. https://doi.org/10.2174/1566524018666180705114131
12. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. https://doi.org/10.1136/bmj.n71
13. Sterne JAC, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS I: a tool for assessing risk of bias in non randomised studies of interventions. BMJ. 2016;355:i4919. https://doi.org/10.1136/bmj.i4919
14. Guyatt GH, Oxman AD, Schünemann HJ, Tugwell P, Alonso Coello P, et al. GRADE guidelines: a new series of articles in the Journal of Clinical Epidemiology. J Clin Epidemiol. 2011;64 (4):380 382. https://doi.org/10.1016/j.jclinepi.2010.09.011
15. Chu SH, Lee MK, Ahn KY, Im JA, Park MS, Lee DC, Jeon JY, Lee JW. Chemerin and adiponectin contribute reciprocally to metabolic syndrome. PLoS One. 2012;7(4):e34710. https://doi.org/10.1371/journal.pone.0034710
16. Chedraui P, Pérez-López FR, Escobar GS, Palla G, Montt-Guevara M, Cecchi E, et al. Circulating leptin, resistin, adiponectin, visfatin, adipsin and ghrelin levels and insulin resistance in postmenopausal women with and without the metabolic syndrome. Maturitas. 2014;79(1):86-90. https://doi.org/10.1016/j.maturitas.2014.06.008
17. Panahi Y, Khalili N, Sahebi E, Namazi S, Simental-Mendía LE, Majeed M, et al. Effects of curcumin on serum adipokine concentrations in patients with metabolic syndrome: a randomized controlled trial. Clin Nutr. 2016;35(2):409-414. https://doi.org/10.1016/j.clnu.2015.03.014
18. Barliana MI, Yolanda PD, Rostinawati T, Ng H, Alfian SD, Abdulah R, et al. Polymorphism of the APM1 gene in subjects with central obesity related to lower high-density lipoprotein cholesterol. Diabetes Metab Syndr Obes. 2019;12:2317-2324. https://doi.org/10.2147/DMSO.S220050
19. Frühbeck G, Catalán V, Rodríguez A, Ramírez B, Becerril S, Salvador J, Colina I, Gómez Ambrosi J. Adiponectin leptin ratio is a functional biomarker of adipose tissue inflammation. Nutrients. 2019;11(2):454. https://doi.org/10.3390/nu11020454
20. Ouerghi N, Ben Fradj MK, Talbi E, Bezrati I, Feki M, Bouassida A. Association of selected adipokines with metabolic syndrome and cardio-metabolic risk factors in young males. Cytokine. 2020;133:155170. https://doi.org/10.1016/j.cyto.2020.155170
21. Sigit FS, Trompet S, Tahapary DL, Sartono E, Willems van Dijk K, Yazdanbakhsh M, et al. The associations of leptin and adiponectin with the metabolic syndrome in an Indonesian and a Dutch population. Nutr Metab Cardiovasc Dis. 2021;31(8):2426-2435. https://doi.org/10.1016/j.numecd.2021.05.012
22. Khademi Z, Hamedi-Shahraki S, Amirkhizi F. Vitamin D insufficiency is associated with inflammation and deregulation of adipokines in patients with metabolic syndrome. BMC Endocr Disord. 2022;22(1):223. https://doi.org/10.1186/s12902-022-01141-0
23. Singh RP, Chauhan K, Tripathi A, Choudhary R. Association of Adipokines and CIMT in Metabolic Syndrome in Western Uttar Pradesh Population: a Cross-Sectional Study. Maedica (Bucur). 2024;19(3):494-501. https://doi.org/10.26574/maedica.2024.19.3.494
24. Cheng D, Ji H, Liu D, Hou X, Jia W, Li H. Adiponectin as a novel biomarker for progression of cardiovascular-kidney-metabolic syndrome—insights from a longitudinal cohort study. Diabetes. 2025;74 https://doi.org/10.2337/db25-2090-LB
25. Lara-Guzmán ÓJ, Arango-González ÁM, Álvarez-Quintero R, Escobar JS, Muñoz-Durango K, Sierra JA. Circulating hs-CRP, IL-18, chemerin, leptin, and adiponectin levels reflect cardiometabolic dysfunction in adults with excess weight. Int J Mol Sci. 2025;26(3):1176. https://doi.org/10.3390/ijms26031176
26. Sinke L, Delerue T, Wilson R, Lu X, Xia Y, Costeira R, et al. DNA methylation of genes involved in lipid metabolism drives adiponectin levels and metabolic disease. Diabetologia. 2026;69(1):127-145. https://doi.org/10.1007/s00125-025-06549-6
27. Pedro WJS, Barbosa Júnior FV, Alves FNB, et al. Role of Adipokines Chemerin, Visfatin, and Omentin in Obesity and Their Inflammatory and Metabolic Implications. Biomedicines. 2025;13(10):2321. https://doi.org/10.3390/biomedicines13102321
28. Yilmaz DT, Gul MA, Capraz M, Demir HD, Tekcan A. Investigation of Serum Visfatin and Chemerin Levels in Type 2 Diabetes and Obesity Patients: Their Potential Role as Clinical and Biomarkers. Biomedicines. 2025;13(11):2619. https://doi.org/10.3390/biomedicines13112619
29. Ouchi N, Parker JL, Lugus JJ, Walsh K. Adipokines in inflammation and metabolic disease. Nat Rev Immunol. 2011;11(2):85–97. https://doi.org/10.1038/nri2921
30. Tan L, Lu X, Jan Danser AH, Verdonk K. The Role of Chemerin in Metabolic and Cardiovascular Disease: A Literature Review of Its Physiology and Pathology from a Nutritional Perspective. Nutrients. 2023;15(13):2878. https://doi.org/10.3390/nu15132878
31. Mancuso P. The role of adipokines in chronic inflammation. Immunotargets Ther. 2016;5:47–56. https://doi.org/10.2147/ITT.S73223
32. Sam S. Differential effect of subcutaneous abdominal and visceral adipose tissue on cardiometabolic risk. Horm Mol Biol Clin Investig. 2018;33(1):1–8. https://doi.org/10.1515/hmbci 2018 0014
33. Strugała A, Jagielski J, Kamel K, Nowaczyk G, Radom M, Figlerowicz M, Urbanowicz A. Virus like particles produced using the Brome mosaic virus recombinant capsid protein expressed in a bacterial system. Int J Mol Sci. 2021;22(6):3098. https://doi.org/10.3390/ijms22063098
34. Chang E, Kim Y. Vitamin D decreases adipocyte lipid storage and increases NAD-SIRT1 pathway in 3T3-L1 adipocytes. Nutrition. 2016;32(6):702–708. https://doi.org/10.1016/j.nut.2015.11.021
35. Kumari B, Yadav UCS. Adipokine Visfatin’s Role in Pathogenesis of Diabesity and Related Metabolic Derangements. Curr Mol Med. 2018;18(2):116–125. https://doi.org/10.2174/1566524018666180705114131
36. Yuan HP, Sun L, Li XH, Che FG, Zhu XQ, Yang F, et al. Association of adiponectin gene +45T>G polymorphism with metabolic syndrome risk and adiponectin level: a meta analysis. Sci Rep. 2016;6:31945. https://doi.org/10.1038/srep31945
37. Wu R, Wang X. Epigenetic regulation of adipose tissue expansion and adipogenesis by N6 methyladenosine. Obes Rev. 2021;22(2): e13124. https://doi.org/10.1111/obr.13124
38. Deng Y, Scherer PE. Adipokines as novel biomarkers and regulators of the metabolic syndrome. Ann N Y Acad Sci. 2010;1212(1):E1–E19. https://doi.org/10.1111/j.1749-6632.2010.05875.x
39. Romacho T, Sánchez-Ferrer CF, Peiró C. Visfatin/Nampt: an adipokine with cardiovascular impact. Mediators Inflamm. 2013;2013:946427. https://doi.org/10.1155/2013/946427
40. Ouwens DM, Sell H, Greulich S, Eckel J. The role of epicardial and perivascular adipose tissue in the pathophysiology of cardiovascular disease. J Cell Mol Med. 2010;14(9):2223–2234. https://doi.org/10.1111/j.1582-4934.2010.01141.x